Yeasen Biotechnology's Commercial HCP Reagent Kit is a simple, highly sensitive, and objective method for HCP detection. Yeasen Biotechnology has independently developed multiple reagent kits, which can sensitively detect residual HCP levels in samples. These reagent kits can be used for optimizing the purification process of biopharmaceuticals, controlling impurities in intermediate processes, and releasing testing of final products. Want to learn more about how Yeasen Biotechnology's HCP reagent kits can improve your biopharmaceutical production? Read the article now!

Concept and Necessity of HCP Detection
Biological products are typically produced using recombinant technology in host cells (such as bacteria, yeast, mammalian, insect, or plant cells), including monoclonal antibodies, recombinant proteins, and vaccines. The manufacturing and purification processes of these products inevitably introduce host cell proteins (HCPs) as impurities, which include both structural proteins of the host cells and growth factors secreted by the host cells, forming a complex mixture with various physicochemical and immunological properties. Despite the use of advanced purification techniques, some host cell proteins may still remain as process-related impurities in intermediate drug products.
These impurities have potential immunogenicity and may enhance the immunogenic response of biopharmaceutical products, affecting their stability and efficacy, thereby posing risks to human safety. Therefore, appropriate monitoring strategies for the removal of HCPs need to be established, and the content of HCPs in the final product needs to be controlled to meet the requirements of drug quality.
Regulatory Requirements for HCPs
The residual content of HCPs in biological products is generally considered a critical quality attribute (CQA) of the product, an important evaluation index for process robustness monitoring, and a key quality control index of the product. Regulatory authorities in various countries have provisions regarding HCPs, requiring analysis and purification of biopharmaceuticals to reduce host cell protein HCPs to acceptable levels. The acceptance level of HCPs will be assessed based on specific circumstances and depends on several factors, including dose, dosing frequency, drug type, and disease severity.
The Chinese Pharmacopoeia (2020 edition) stipulates: For CHO cells, the residual HCP needs to be <0.05% (equivalent to less than 500 ppm); for E. coli, the residual HCP needs to be <0.01%.
The United States Pharmacopeia USP <1132> chapter specifies: A highly sensitive method should be used to detect HCPs in drugs, and the content should be below the detection limit (usually less than 100 ppm, i.e., the HCP content in 1 mg total protein should be less than 100 ng, or <0.01%).
The European Pharmacopoeia EP 2.6.34 specifies: In biological products, the content of HCPs should be less than 0.1%.
The International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) guidelines: ICH Q6B states that sensitive and validated methods should be adopted to monitor residual HCPs, with the residual amount typically required to be less than 100 ppm.
It can be seen that regulatory agencies have highly consistent requirements for HCPs. The process development of biological products must undergo HCP detection, demonstrate that the purification process can reduce HCPs to safe levels, and the recommended limits for residual content in the final product are below 0.01% in both China and the United States.
HCP Detection Methods
Enzyme-Linked Immunosorbent Assay (ELISA) is currently the most commonly used method for HCP detection. The host protein residue detection methods mentioned in Chapters 3412/3413/3414 of the 2020 edition of the Chinese Pharmacopoeia are all ELISA methods.
The United States Pharmacopeia USP <1132> chapter also mentions that methods such as electrophoresis, ELISA, and LC-MS/MS can be used to detect HCP content, and ELISA is recommended as the preferred method for HCP detection due to its ease of operation, speed, high sensitivity, and high throughput.
ELISA:
Purpose: Detects the total amount of proteins in HCPs, can be used for product development and process control, and is currently a common method for detecting HCPs.
Advantages: High sensitivity and specificity; High throughput and automation capability; Quantitative; Easy and fast operation.
Disadvantages: Requires specific antibodies; Cannot specifically identify and evaluate individual HCPs; Method development is complex, requiring special antigen preparation; Many influencing factors for the method, HCP coverage needs to be evaluated; Cannot identify species.

Western Blotting:
Purpose: Used for consistent screening of large numbers of samples and detecting unknown proteins that react with anti-HCP antibodies. Suitable for detecting HCPs and providing approximate information on the relative molecular weight of HCPs.
Advantages: Specificity; Results are visually intuitive: visualizes differences in protein size and expression levels; Can detect and quantify specific HCPs.
Disadvantages: Semi-quantitative; Limited to specific proteins: can only detect HCPs bound to antibodies; Not sensitive enough for complex protein mixtures; Protein SDS denaturation may lead to loss of conformational epitopes; Sensitivity depends on the quality of polyclonal antibodies.
Two-Dimensional Gel Electrophoresis (2-DE):

Purpose: Commonly used for upstream or downstream process development and characterization, it can distinguish different HCPs on a single gel.
Advantages: No need for Western blotting, avoiding transfer issues; Can separate trace amounts of HCP impurities from the product; Provides approximate information on relative molecular weight and isoelectric point.
Disadvantages: Excessive protein may mask HCP spots; The distribution of HCPs in the sample can be observed, but cannot be quantified.

Mass Spectrometry Analysis:
Purpose: Identifies individual HCPs, providing accurate identification and quantification information for host cell proteins.
Advantages: High resolution and accuracy; Identifies and monitors individual HCPs; Provides comprehensive spectra of HCPs; Can cover a wide range of proteins; Can identify HCP species.
Disadvantages: High equipment and technical requirements; Complex operation; High cost; Low method throughput; Complex sample pretreatment; Potential matrix effects; Requires specialized data analysis capabilities.
ELISA is the recommended method for detecting residual HCPs in biological products by pharmacopoeias in various countries, capable of determining the total amount of HCPs. However, it has limitations in identifying the types and abundances of HCPs and needs to be supplemented with other methods. Each detection method has its advantages and disadvantages, and in practical applications, selection can be made based on experimental purposes, sample characteristics, accuracy, and operational convenience, among other factors, combined with the use of different methods to obtain more comprehensive and accurate results.
In addition, the United States Pharmacopeia USP <1132> and the European Pharmacopoeia EP 2.6.34. HOST-CELL PROTEIN ASSAYS indicate that different ELISA reagents should be used for HCP detection at different stages of product development, categorizing HCP detection methods into commercial reagents, product/process-specific methods, and platform methods.
USP <1132> mentions that in the absence of platform methods, commercial reagents can be used in preclinical, Phase I, and Phase II clinical trials; in Phase III clinical trials/process validation and post-marketing product, due to limitations such as insufficient antibody coverage of commercial general HCP detection reagents, consideration should be given to factors such as cell type and process specificity, using platform methods or product/process-specific methods for upstream process development.
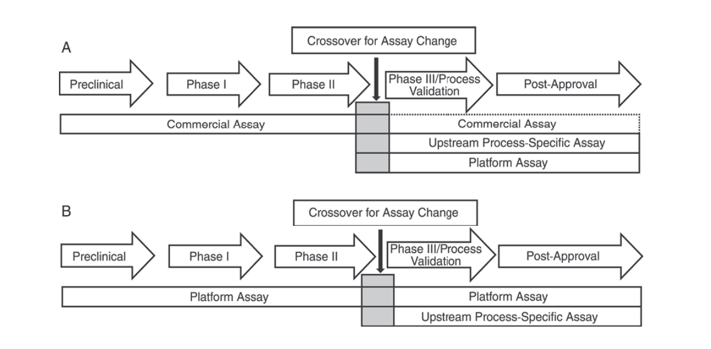
Figure 1. In the USP <1132>, different stages of product development are suggested to use specific methods for detecting HCPs.
|
Method |
Commercial Reagent Kit |
Product/Process-Specific Detection Kit |
Platform Detection Kit |
|
Availability |
Readily available
|
Requires preparation of antigens, including cloning of host cell lines
|
Requires preparation of antigens using nude or empty vector cell lines
|
|
Development |
Requires minimal preparation |
Detection development including antigen characterization, immunization, and confirmation typically takes at least 6 months |
Detection development including antigen characterization, immunization, and confirmation typically takes at least 9 months |
|
Diversity |
Limited to the most common expression systems |
Various types of expression systems can be used |
Various types of expression systems can be used |
|
Specificity |
Specificity may be poor, with potential undetected HCPs
|
Specificity is usually enhanced, with antibody coverage targeting specific HCP patterns |
Compared to commercial detection methods, specificity is improved, understanding of detection performance is enhanced, and coverage of HCP types is also increased |
|
Cost |
Initial prices are relatively cheap, but development costs will quickly rise for the entire downstream purification process |
Initial costs are higher, but the breakeven point is typically at the value of 100 sets of reagent kits |
Due to its applicability to multiple products, it has significant cost advantages |
|
Dependency |
Dependent on a specific supplier, reagents, and control schemes are in a "black box" state. |
Has comprehensive control over all reagents and buffers, can be adjusted at any time |
Has complete ownership and control over all reagents, reducing risks in the supply chain and batch comparability |
|
Regulatory Requirements |
Not suitable for supporting marketing authorization applications |
Applicable for supporting the entire process from early development to the marketing of licensed products |
Applicable for supporting early development until marketing authorization |
Table 1. Comparison of Different HCP Reagent Kit Types
Yeasen Biotech Customized HCP Assay Development Service
Given the significance of HCP detection, Yeasen Biotech relies on a high-performance monoclonal antibody development platform to establish protein antibody critical raw material development platforms and an independent laboratory for immune detection product development, equipped with advanced research instruments such as enzyme immunoassay analyzers and 2D-WB bidirectional gel electrophoresis-immunoblotting systems. The development platform is highly mature and capable of rapidly customizing HCP assay kits for various host cell residues including mammalian cells, insect cells (such as Sf9), bacteria, and yeast.
Customization Timeline
The customization service for host cell residue protein detection kits typically takes 6 to 10 months.
Antigen-Antibody Preparation: Determining HCP antigens, establishing HCP reference standards, devising immune pathways and strategies, preparing HCP antibodies, etc., typically takes 4 to 6 months.
Assay Kit Development: Antibody optimization, coverage analysis, assay kit method validation, etc., typically takes 2 to 4 months.
Yeasen Biotech HCP Antibody Coverage Verification Service
It is known that ELISA is the gold standard for detecting residual HCP in the industry, but the accuracy of this method depends on the coverage of multi-clone antibodies to HCP. The United States Pharmacopeia and European Pharmacopeia mention immunopurification and 2D-WB as recommended methods for characterizing HCP ELISA antibodies and their coverage.
HCP Antibody Coverage Verification Service
2D-WB first separates proteins based on size and charge using two-dimensional gel electrophoresis, then transfers these proteins to a membrane for incubation with HCP antibodies for protein imprint detection.
We has a full set of 2D-WB instrumentation and extensive laboratory experience to provide antibody coverage verification services for HCP samples from host cells such as CHO, E.coli, HEK293, etc.
Related Product:
|
Product |
Catalog No. | Size |
| 36712ES | 48T/96T | |
|
36713ES |
48T/96T | |
|
CHO HCP ELISA kit (CHO-K1) |
36714ES |
48T/96T |
| E.coli HCP ELISA Kit (Plasmid) | 36721ES | 48T/96T |
